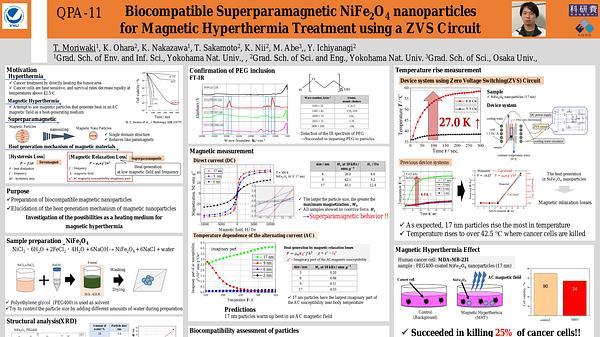
Premium content
Access to this content requires a subscription. You must be a premium user to view this content.

Would you like to see your presentation here, made available to a global audience of researchers?
Add your own presentation or have us affordably record your next conference.
The use of magnetic nanoparticles for cancer cell destruction is widely known due to their applications such as magnetic hyperthermia and magneto mechanical actuation
(MMA). Previously, we presented our results regarding a new type of magnetic particles (MPs), i.e. Fe-Cr-Nb-B MPs, prepared by wet milling of superferromagnetic precursor glassy
ribbons, designed for cancer treatment by MMA 1. These MPs have an important shape anisotropy and a large saturation magnetization, which generates an improved torque in a rotating
magnetic field, producing important damages on the cellular viability of cancer cells. Inspired by these findings, we tested the possibility of using magnetic nanowires (NWs) that have a
higher shape anisotropy. Furthermore, the Fe-Co alloy that we propose has high saturation magnetization values. We prepared Fe64.5Co35.5 NWs with lengths of 4 microns by electrolytic
deposition in alumina membranes with a pore diameter of 200 nm.
The Fe-Cr-Nb-B MPs and Fe-Co nanowires were tested on human osteosarcoma cells (HOS) using various concentrations of particles and found that both are biocompatible in vitro.
Subsequently, we studied the effect of MMA in a rotating 80 Oe magnetic field and frequency of 2 Hz on the viability of HOS. We used MPs and NW with different concentrations, and
exposure time of 30 minutes, applied 24 hours after the introduction of the MPs and NWs into cell culture media. The results indicated that with a concentration of 1 mg/ml of MPs and 0.1
mg/ml of NWs, respectively, the cell viability decreased to about 6-7%. The concentration of NWs needed to induce cancer cell death is 10 times lower than the one needed for MPs. It is
worth mentioning that although NWs are more efficient when using low quantities, due to their higher magnetization and anisotropy, the magnetic nanoparticles are better internalized and
metabolized by cells. Therefore, MPs can be better targeted in tissues harder to reach than NWs. The latter are also harder to control, being more suitable for superficially-developed cancer
tissues.
Supported by (UEFISCDI) Contract no. PCE20/2021 (PN-III-P4-ID-PCE-2020-2381).
References:
1. H. Chiriac et al. Sci. Rep., 8.11538 (2018)