Premium content
Access to this content requires a subscription. You must be a premium user to view this content.

Would you like to see your presentation here, made available to a global audience of researchers?
Add your own presentation or have us affordably record your next conference.
Currently, the treatment of suppression of cytokine storm in severe COVID-19 patients includes cytokine receptor suppression and depletion of specific immune cells in
addition to steroid treatment. Monoclonal antibody therapy is also one of the methods for depleting specific immune cells. It is a monoclonal antibody therapy against CD3, one of the
important signaling molecules for T-cell receptor activation. Among others, T cells secrete interferon gamma (IFN-γ), a pro-inflammatory cytokine responsible for inducing inflammation.
Therefore, monoclonal antibody therapy againstis a therapy that specifically depletes IFN-γ-secreting T cells. However, intravenous injection of an anti-CD3 monoclonal antibody targeting
all T cells induces significant immunosuppression and has side effects such as high fever and chills. The purpose of this study is to prepare an optimized liquid phase as a CD3 antibodymagnetic
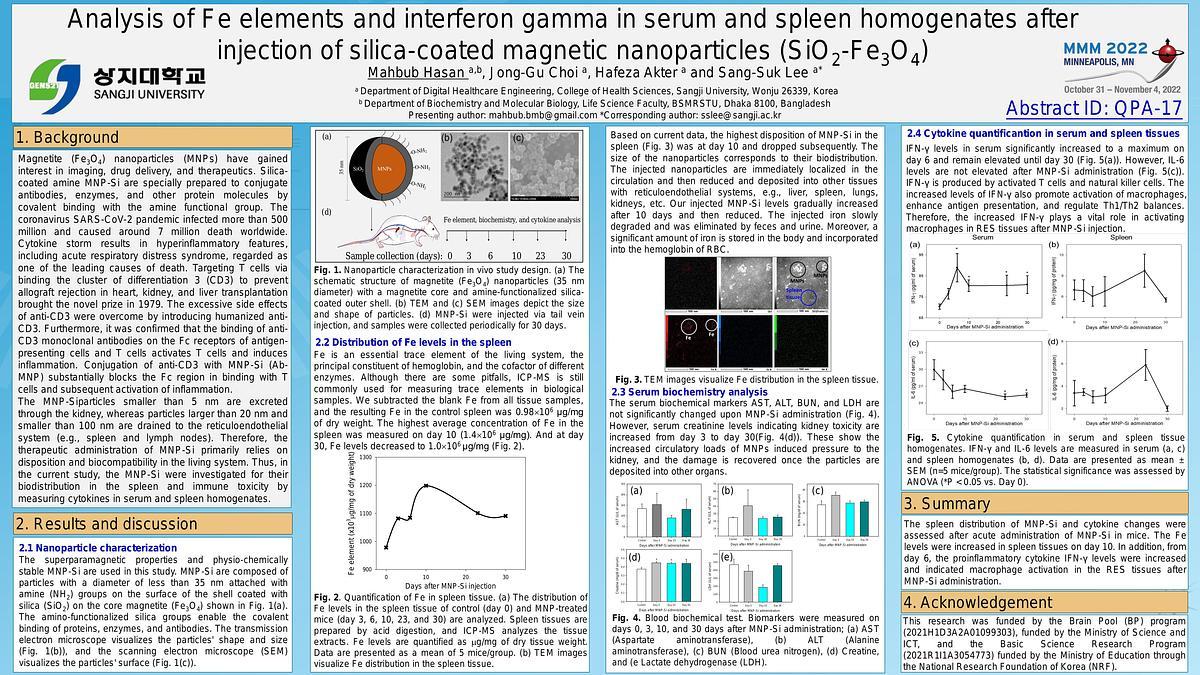
nanoparticle (Ab-MNP) conjugate to inhibit the overactivation of T cells. In current study, we aimed to check distribution of Fe after acute admistration of silica-conjugated
amine nanoparticles delivered by intravenous injection. Besides, the IFN- γ levels in serum and spleen homogenates will be analyzed. Finally, we will develop film-type microneedle patch
using pulse-type magnetic stimulation to control the amount of liquid passing through the skin. The reason for choosing the spleen from the mouse organ is that it stores MNPs with a
particle size of 50 nm administered in vivo. Therefore, the result is a difference in the concentration of Fe elements, which was 1.89×103 mg/kg in the spleen of a control mouse not
administered with MNPs, whereas increases significantly to 1.93×103 mg/kg in that of a mouse administered with MNPs as shown in Fig. 1(a) and 1(b). IFN-γ level in spleen tissue and
serum is increased after 10 days. Further, time kinetic analysis of biochemical and immunological parameters are required to confirm its suitability in bio-administration.
References:
1 N. S. Remya, S. Syama, A. Sabareeswaran, and P. V. Mohanan, Toxicity, toxicokinetics and biodistribution of dextran stabilized Iron oxide Nanoparticles for biomedical
applications, International Journal of Pharmaceutics 511, 586-598 (2016).
2 F. Gao, An Overview of Surface-Functionalized Magnetic Nanoparticles: Preparation and Application for Wastewater Treatment, Chemistry Select 4, 6805-6811 (2019).<br<
3 R. Q. Cron, R. Caricchio, and W. W. Chatham, Calming the cytokine storm in COVID-19, Nature Medicine 27, 1674–1675 (2021).
4 C. Kuhn and H. L. Weiner, Therapeutic anti-CD3 monoclonal antibodies: from bench to bedside, Immunotherapy 8, 889–906 (2016).