Premium content
Access to this content requires a subscription. You must be a premium user to view this content.

Would you like to see your presentation here, made available to a global audience of researchers?
Add your own presentation or have us affordably record your next conference.
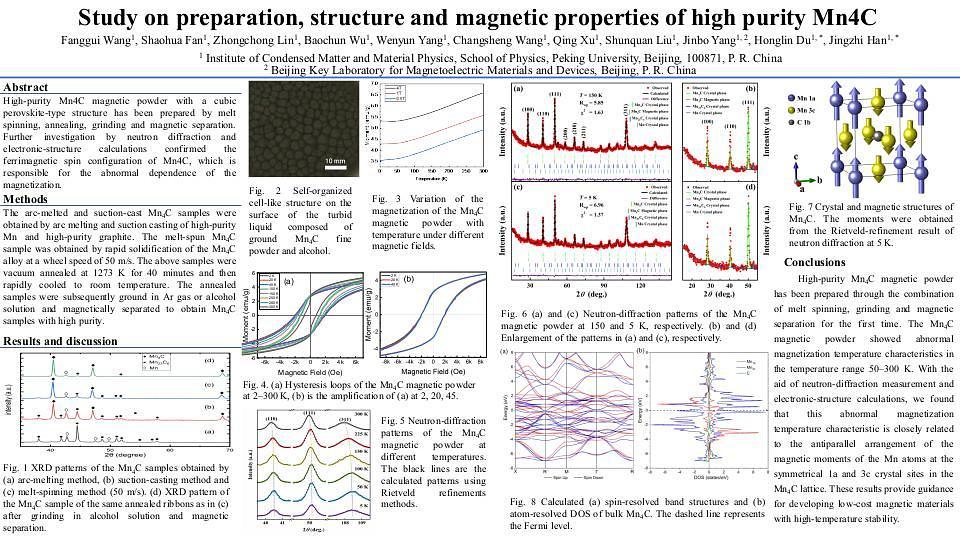
The magnetization of Mn4C with face centered cubic structure increases with the increase of temperature within a certain temperature range, which means that Mn4C may be applied to the field requiring high working temperature of magnetic materials through further research and development. Unfortunately, Mn4C is a metastable phase 1-3, so it is difficult to synthesize high-purity Mn4C.
High-purity Mn4C powders was prepared by melt spinning, grinding and magnetic separation in this paper. It is found that high cooling rate is beneficial to obtain high-purity Mn4C material. Moreover, compared with argon gas as the grinding medium, grinding Mn4C melt-spun strips in alcohol solution is not only conducive to refining the size of the ground particles, but also conducive to improving the dispersion of the particles. Therefore, Mn4C powders with higher purity can be obtained after magnetic separation (see Fig. 1).
The magnetization of Mn4C powders remained unchanged with the increase of temperature in the range of 2 K -50 K, but increased with the increase of temperature in the range of 50 K to 300 K (see Fig. 2). This may be because there is a spin glass state in Mn4C powders. At low temperature, the spins are frozen, but the frozen spins are gradually thawed with the increase of temperature. Further investigation is in progress.
Crystal and magnetic structure of Mn4C powders was investigated by variable temperature neutron diffraction technique. The magnetization change of Mn4C with temperature should be mainly due to its own magnetic phase transition, and is closely related to the antiparallel arrangement of the magnetic moments of Mn atoms at the symmetrical crystal positions 1a and 3c in the Mn4C lattice. The total magnetic moment of crystal cells increases with the increase of temperature.
References:
1. Si, PZ, Qian, HD, Ge, HL,et al. Applied physics letters, 2018, 112(19):192407.
2.Park ,J, Qian, HD, Si, PZ, et al. Journal of magnetism and magnetic materials, 2021, 527:167765.
3. Tagawa, Y and Motizuki, K. Journal of physics-condensed matter, 1991, 3(12): 1753-1761

Fig.1 XRD patterns of Mn4C obtained by (a) arc melting, (b) Suction casting, (c) melt spinning technology (50m/s),and (d) grinding and magnetic separation of the same strips as (c) in alcohol solution. (a) (b) and (c) are both heat treated and ground and magnetically separated in argon.

Fig.2 (a) Variation of magnetization of Mn4C powder with temperature under different magnetic field; (b) Hysteresis loops of Mn4C powders at 2K-300K

