Premium content
Access to this content requires a subscription. You must be a premium user to view this content.

Would you like to see your presentation here, made available to a global audience of researchers?
Add your own presentation or have us affordably record your next conference.
Nowdays, Nd-Fe-B magnetic powder is usually prepared by the strip casting (SC), hydrogen decrepitation (HD) and jet milling (JM), so as to obtain high performance. However, due to the harsh hydrogen absorption conditions of Sm-Co alloy, the HD is rarely used for its powder preparation, and there is no research on the hydrogenation behavior of SC flake. Therefore, it is urgent to introduce a new powder preparation process for mass production. In this study, the hydrogen absorption behavior of Sm(CoFeCuZr)z SC flake was systematically investigated.
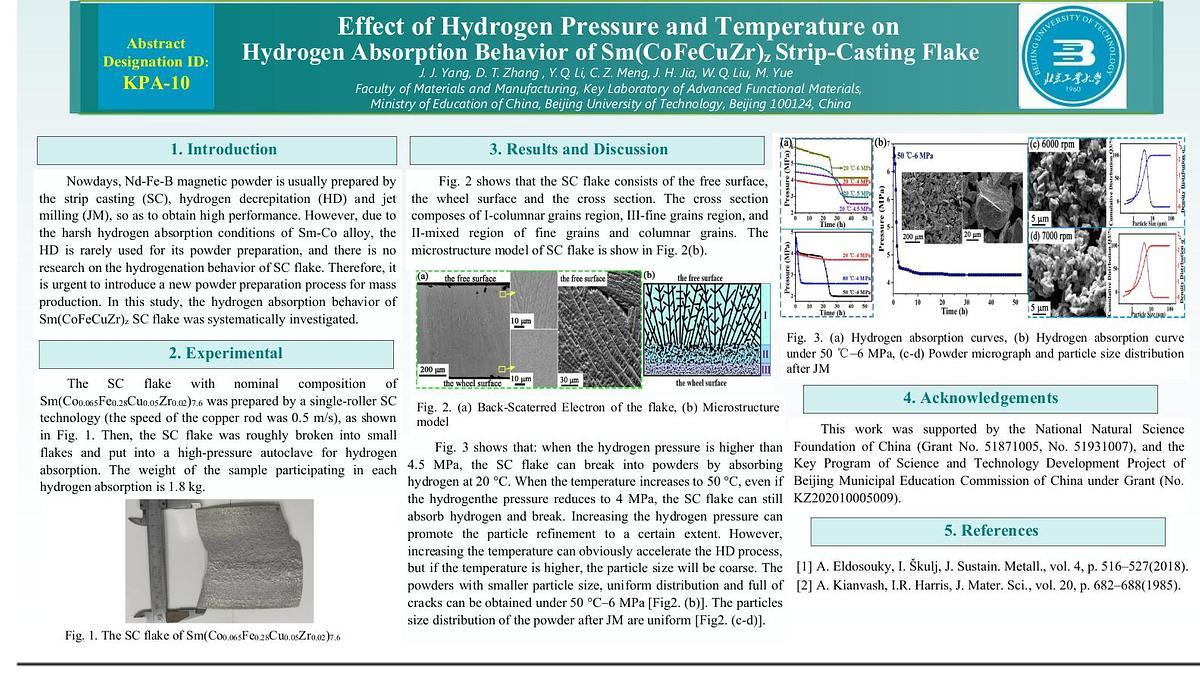
The SC flake with nominal composition of Sm(Co0.065Fe0.28Cu0.05Zr0.02)7.6 was prepared by a single-roller SC technology. The SC flake composes of the free surface, wheel surface and cross section. The cross section consists of I-columnar grains region, III-fine grains region, and II-mixed region of fine grains and columnar grains Fig1. (a-b). The weight of each sample involved in hydrogen absorption is 1.8 kg. The hydrogen absorption curves show that: when the hydrogen pressure is higher than 4.5MPa, the SC flake can break into powders by absorbing hydrogen at 20 °C. When the temperature increases to 50 °C, even if the hydrogenthe pressure reduces to 4 MPa, the SC flake can still absorb hydrogen and break. Increasing the hydrogen pressure can promote the particle refinement to a certain extent. However, increasing the temperature can obviously accelerate the HD process, but if the temperature is higher, the particle size will be coarse. The powders with small particle size, uniform distribution and full of cracks can be obtained under 50 °C-6 MPa Fig2. (b), which is conducive to the subsequent preparation of fine powder. The particles size distribution of the powder after JM are uniform Fig2. (c-d). In conclusion, the hydrogen pressure and temperature are the key factors for HD process. The SC, HD and JM can be used as a new powder preparation process, which is expected to prepare high-performance Sm(CoFeCuZr)z magnets in the future.
References:
1 A. Eldosouky, I. Škulj, J. Sustain. Metall., vol. 4, p. 516–527(2018).
2 A. Kianvash, I.R. Harris, J. Mater. Sci., vol. 20, p. 682–688(1985).

Fig. 1. (a) Back-Scaterred Electron of the flake, (b) Microstructure model

Fig. 2. (a) Hydrogen absorption curves, (b) Hydrogen absorption curve under 50 °C-6 MPa, (c-d) Powder micrograph and particle size distribution after JM

